Copper (Cu) contamination can cause Copper Contamination Cracking (CCC)on austenitic stainless steels weldment when the surface at or near HAZ (Heat Affected Zone) get contaminated and the cracks will be resulted from LME (Liquid Metal Embrittlement)and intergarnular cracking
Cu as an alloying element improves the corrosion resistant of stainless steels in reducing acids like sulfuric and phosphoric acids. In intermediate concentration (25 to 70%) and concentrated (70 to 99+%) sulfuric acid, super-austenitic stainless steel grade 904L (contains 1.5% Cu) has a very good resistant over the Cu free grades. The presence of copper in stainless steels has longbeen known to improve their resistance to sulfuric acid solutions.
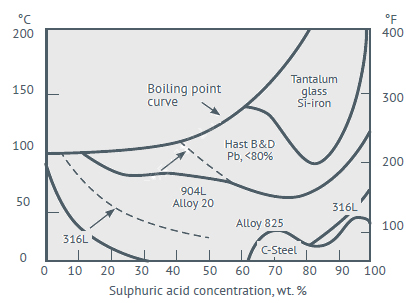
0.5 mm/y (20 mpy) Iso-corrosion curves for selected alloys in Sulfuric acid service
The issue of CCC can result from surface contamination where the temperature at the HAZ or during the application of annealing heat treatment where the temperature exceeds the melting temperature of Cu (~1083 °C) while 304L stainless melts over a temperaturerange of about 1400°C to 1450°C.The molten copper wets very rapidly along the grain boundariesof the HAZ as a liquid film. As the weld cools and shrinks, it imposes a tensile stresswhich the copper-rich liquid film cannot withstand. Then cracks form along thesegrain boundaries in the HAZ.
The usual scenario when this occurs is thatcopper is rubbed on the stainless surface, as for example, by sliding the stainlessagainst a copper chill bar on a seamer. Microscopic bits of copper rub off and adhere tothe surface of the stainless.
A copper backing bar is often used when welding sheet material. It is important to avoid rubbing the stainless steel against the Cu which can transfer Cu to the stainless steel surface.
So, it should be abundantly clear that the surface of austenitic stainless steels such as304L and 316L should be protected against having copper rubbed on the surface.
Direct contact, such as with a chill bar or backup bar or alignment clamp, can be acceptableas long as rubbing is avoided.
Also, stainless steel must not be stored in contact with, or in the same area, as galvanized steel or copper. Surface contamination by Copper, Zinc, or other low-melting metals can embrittle stainless steels

Inspection and Identification of CCC
- PT can be used to detect the surface cracks initiated by CCC at the HAZ
- Metallography can be used to detect the intergranular cracks in HAZ and to detect presence of copper at the grain boundaries in as-polished conditions.
References:
- WJ_1982; Copper-Contamination Cracking: Cracking Mechanism and Crack Inhibitors
- WJ 1978; Liquid-Metal Embrittlement of the Heat-Affected Zone by Copper Contamination
- Nidi, Welding of stainless steels and other joining methods
- Nidi; Practical Guidelines for the fabrication of Austenitic Stainless steels
- AWS; Welding of stainless steels- Questions and answers
- MTI Publication MS-1 second edition, “Materials Selector for Hazardous Chemicals — Sulfuric Acid”, Materials Technology Institute of the Chemical Process Industries, Inc., St. Louis, MO (2005).
- Nidi, Alloy selection for service in sulphuric acid
Written By:

Baher Elsheikh, Mechanical engineer has 20 years of experience in oil and gas and petrochemical industry, particularly in the engineering, reliability, construction and troubleshooting of pressure equipment and fired heaters. He worked in refinery, petrochemical and fertilizer plants as a senior mechanical engineer specialized in static equipment.