X-Ray Fluorescence (XRF) Technique
Iron Ore Analysis Using Portable XRF and XRD
Published on 2nd June 2023

image credit @Olympus
Olympus portable X-ray fluorescence (pXRF) and X-ray diffraction (pXRD) analyzers play an important role in the exploration and mining of iron ores and deposits. For example, the Vanta™ handheld XRF analyzer can accurately measure the full range of iron (Fe) concentrations in samples with a wide variety of deposit types while the BTX™ III and TERRA™ II mobile XRD analyzers can identify and quantify various iron phases down to 1% concentration by weight.
Almost 2.5 billion tons of iron ore were mined in 2020, making iron ore central to the world economy.1 In fact, mining analysts have stated that iron ore is “more integral to the global economy than any other commodity, except perhaps oil.”2 Vanta pXRF analyzers can quantify iron concentrations in ores and deposits in the field. This analysis can be performed in 30 seconds or less without the need for indirect detection, such as the use of pathfinder elements, or complicated sample preparation. Similarly, Olympus pXRD instruments can quantify a wide variety of iron minerals in ores and deposits down to 1% in the field without complicated sample preparation or external hardware or infrastructure.
Vanta™ pXRF Performance of Iron and Associated Elements
Most iron ore is easy to identify based on its natural abundance (fourth most abundant element on Earth) and high concentrations of iron (up to 75% Fe by weight). However, many deposits contain other economically important materials and elements that either aid or hinder the mining or processing steps of iron production, as shown in the table below.
| Element of Interest | Importance |
| Aluminum (Al) | Increases the viscosity of iron slag |
| Silicon (Si) | Used in production due to its decreased brittleness and ease of casting |
| Phosphorous (P) | Varies the mechanical and thermal properties of iron |
| Sulfur (S) | Greatly increases the brittleness of metallic iron, making it less workable |
| Calcium (Ca) | Can be added as lime during iron production to remove other impurities |
The ability to determine the concentration of iron and other important elements is critical for effective pXRF analysis. In a matter of seconds, Vanta pXRF analyzers can provide accurate concentrations of these elements and others with simple in-field sample preparation. These fast, accurate results are useful for making real-time decisions at a worksite, helping to eliminate the expensive downtime—sometimes up to days—involved in incorrect exploration or sending samples to a lab.
The data below illustrates the standard performance of a Vanta pXRF analyzer on a range of certified reference materials (CRMs) in various kits supplied by OREASOREAS (Ore Research and Exploration Assay Standards). Exceptional accuracy and precision between the CRM data and calculated concentrations from the Vanta pXRF analyzer demonstrate its ability to provide excellent, high-quality data on iron ores and deposits.3
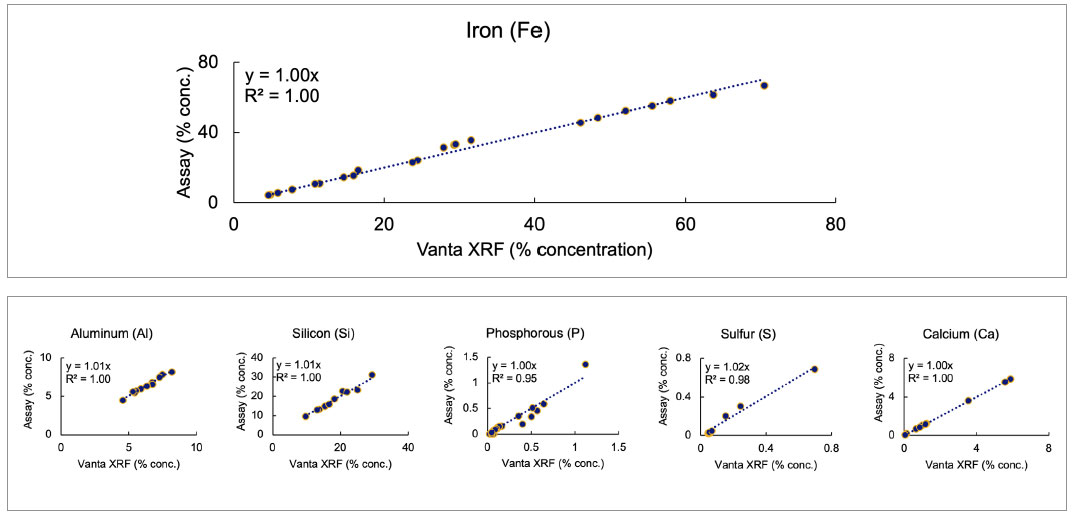
Figure 1. Vanta pXRF performance of iron (Fe) and other critical elements compared to standards using various iron ore kits supplied by OREAS.
The Vanta™ pXRF analyzer demonstrated high performance over various ore samples, including hematitic breccia, hematitic banded iron formations (BIF or taconite), ferruginous soil, lateritic soil, and magnetite skarn—all matrices that commonly contain iron ore. The graphs show that the Vanta analyzer provides accurate, precise results across all matrices and concentration levels, as indicated by the slopes and R2 being close to 1.
These iron ore deposits and matrix types can be found across the globe. Australia, Brazil, China, and India are the four largest iron ore producers in the world, responsible for almost 80% of the worldwide production of usable iron ore.1 Some of the world’s largest mines include the Carajás Mine in Brazil, the Zanaga Project in the Republic of Congo, the RTZ Iron Ore Mine in the Hamersley Basin in Australia, and the Sishen Mine in South Africa. While these large mines are common in Brazil, Western Australia, and Sub-Saharan Africa, there are iron ore mines all over the world, as shown in the map below.
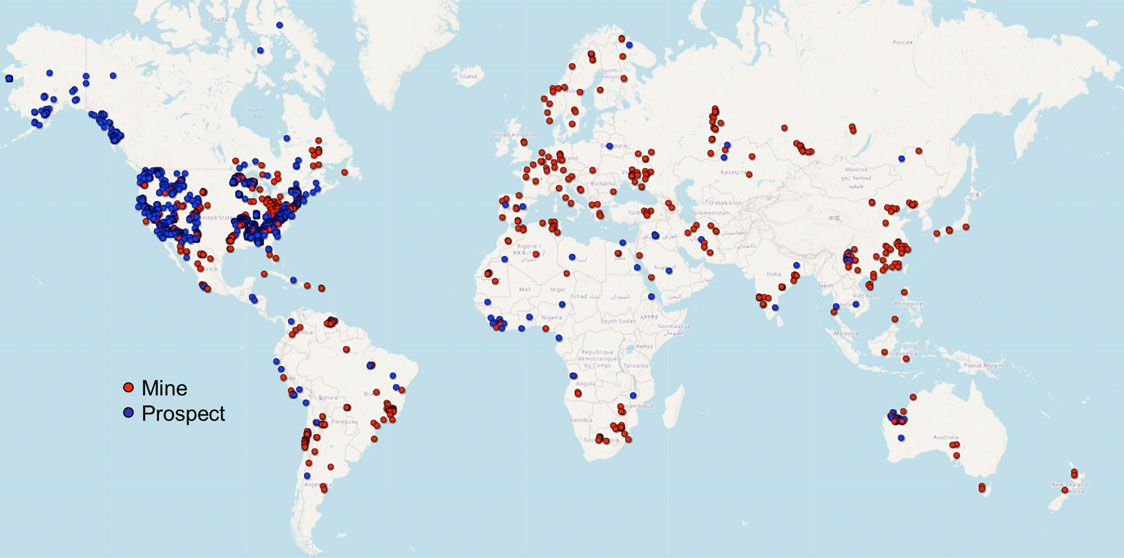
Figure 2. Map of active iron mines and prospects worldwide. Source: United State Geological Survey
Much data is available that shows how the Vanta™ pXRF analyzer can produce excellent quality data on partially and unprepared samples for a wide range of commodities. View some of this data here:
- Handheld XRF for Soil Surveys: Geochemistry of Rock Outcrops, Soils, and Sediments
- Handheld XRF in Exploration Drilling: Reverse Circulation/Rotary Air Blast and Diamond Core
- Portable XRF for Gold (Au) and Au Pathfinders for Mineral Exploration and Ore Body Vectoring
- Portable XRF for Analysis of Copper Gold Porphyry Deposits
- Portable XRF for Rare-Earth Element Identification and Exploration
Iron Ore Mineralization
Most iron ore mined today comes from banded iron formations (BIF), magnetite ores, hematite ores, and magmatic magnetite deposits. These ores and deposits can contain upwards of 70% iron by weight and are located all over the world. Most remaining materials are silicates and other oxides. The most common of these iron oxides are wüstite (FeO), hematite (Fe2O3), and magnetite (Fe3O4).
While calculating the elemental concentrations, the Vanta pXRF analyzer can also calculate these mineral and compound concentrations in real time. With the easy-to-use interface, the user can set the instrument to report the calculated concentrations as compounds, as demonstrated in Figure 3 for a BIF reference standard. The Vanta software performs this calculation, taking only a few seconds to switch between elemental and compound reporting.
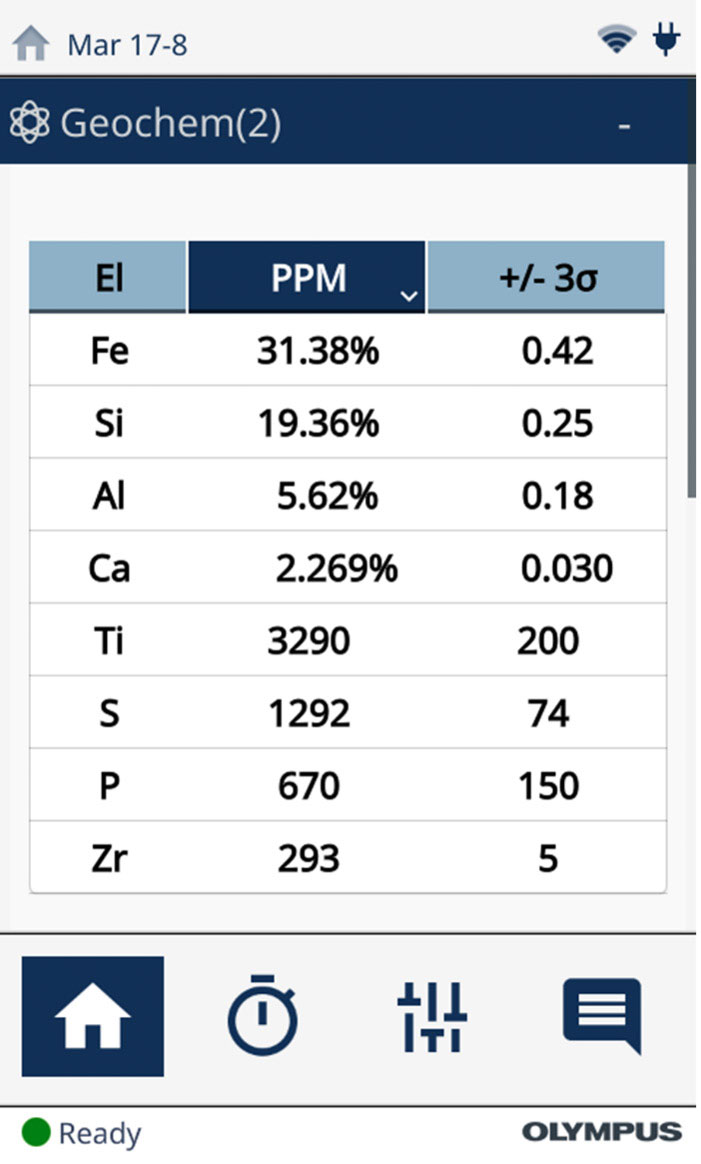
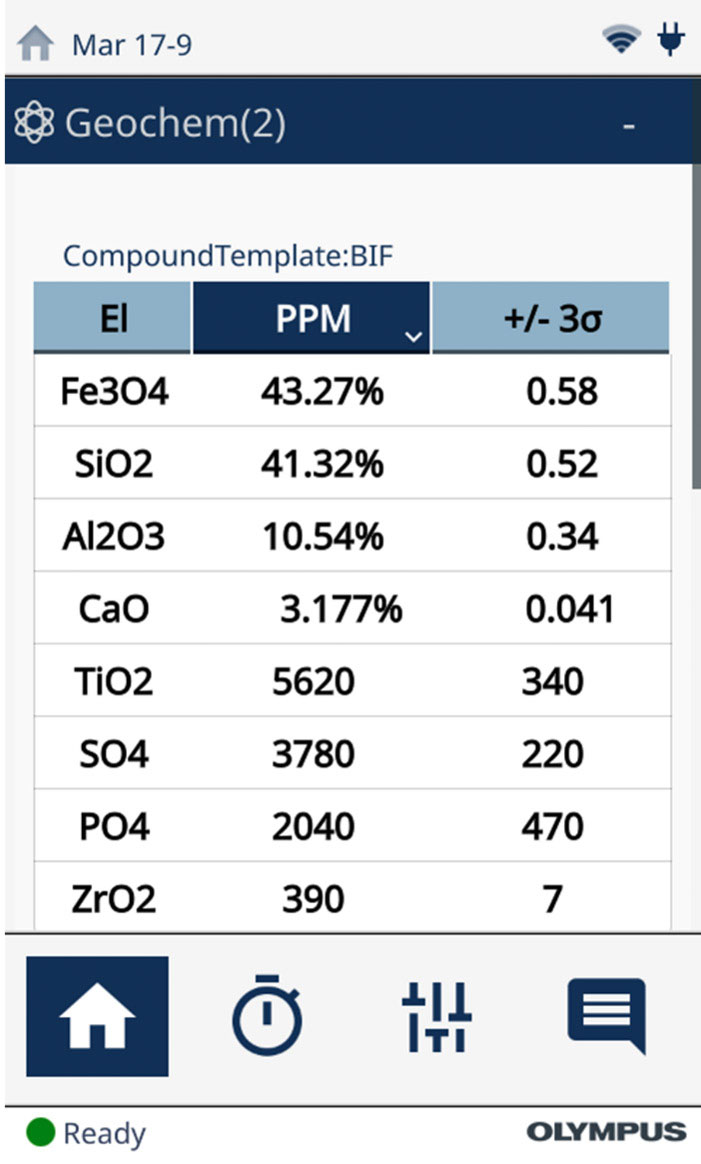
Figure 3. Scan of an iron ore sample without (left) and with (right) the Vanta analyzer’s compound function enabled
Iron Grade Exploration and Process Control Using Olympus pXRD
The TERRA™ II XRD analyzer is the successor to the world’s first commercial battery-operated, portable XRD instrument. Just like the compact benchtop BTX™ III analyzer, this pXRD instrument can identify and quantify various iron phases in a single sample. Technicians and scientists can use this tool to determine the grade, purity, and quantity of specific types of iron ore and iron-containing minerals in real time at the mine or processing facility. Olympus pXRD analyzers can identify all mineral phases in the field in real time, regardless of chemistry and orientation.
As shown in the diffractogram below, the TERRA II and BTX III XRD analyzers can quickly identify various phases in an iron ore sample. This synthetic sample was prepared to demonstrate the instrument’s ability to differentiate and quantify between the various phases of metallic iron and iron oxide accurately and precisely. Each component of the sample can be identified and quantified in real-time using the integrated SwiftMin® automated phase ID and quantitative software or with standalone software packages such as XPowder (included). These options make it even easier and faster to quantify phase information and mineralogy.
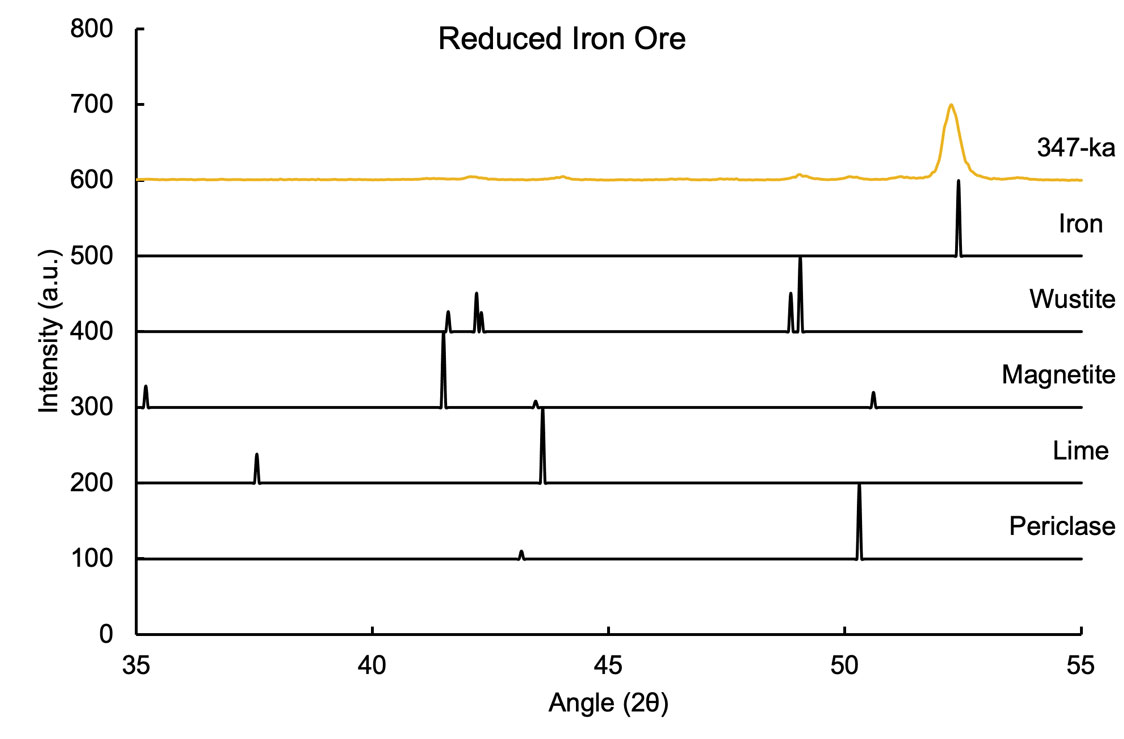
Figure 4. Diffractogram of reduced iron oxide and peak locations with relative peak heights of constituent phases, along with concentrations obtained through the reference intensity ratio (RIR) technique using XPowder software
| Iron | Wustite | Magnetite | Lime | Periclase | Amorphous | |
| Sample | 46.4 | 12.1 | 2.9 | 15.8 | 18.7 | 4.1 |
| Chemistry | Fe | FeO | Fe3O4 | CaO | MgO |
This iron ore was used to demonstrate the Olympus pXRD analyzer’s ability to distinguish various iron-containing phases and any impurities in the sample. While pXRF can only quantify the total concentration of iron in the sample, pXRD can distinguish between various types. In this example, the sample has metallic iron (Fe), along with two forms of iron oxide, wüstite (FeO), and magnetite (Fe3O4). The materials also have some lime impurities left over from processing, along with periclase, a magnesium oxide that occurs commonly in nature with wüstite to form the material ferropericlase.
One inexpensive way to process and purify iron ores is through the production of direct reduced iron (DRI), also called sponge iron. The direct reduction process is a solid-state processing method of reducing various iron oxides to metallic iron at lower temperatures (well below 1,200 °F). Due to this decreased temperature, steel produced using this technique is more energy efficient than other production techniques.4 Determining the concentration of the various phases during DRI processing can improve efficiency and cut costs for iron production. Many processing facilities either use wet chemistry techniques, which are not as accurate at determining phase concentrations, or external labs, which are expensive and time-consuming. The BTX™ III and TERRA™ II analyzers offer easy sample preparation and fast analysis times for high-throughput testing, enabling real-time decision-making and quality control. Olympus has done some previous work on QA/QC of iron ore using pXRD.
Olympus pXRD technology has many benefits, including:
- Fast acquisition: go from sample prep to analysis in minutes
- Virtually maintenance-free: runs without the need for compressed gas, water cooling, a secondary chiller, or an external transformer to decrease downtime and costs
- Easy sample prep: simple mortar-and-pestle process can be done by operators of all experience levels in the field quickly and without complex equipment
- Small sample holder: only requires 15 mg of material compared to the large batch required for conventional XRD